 Metallic elements lose their valence electrons (as bolded in the table) to achieve the noble gas configuration, hence forming positively charged ions. They do not include the transition metals between group II and III. Main-group elements are those in groups I to VII, and group 0 (noble gases). Given that metallic ions lose electrons while non-metallic ions gain electrons to achieve the noble gas electronic configuration, we can use this as a rule of thumb to predict the charges of main-group elements. Amongst them, metallic ones are in blue, while non-metallic ones are in red. Predicting the charges of common ions in an ionic compound Main-group elements in the first four periods are those highlighted. These behaviors merge into each other seamlessly in various circumstances, so that there is no clear line to be drawn between them.7. This attraction may be seen as the result of different behaviors of the outermost or valence electrons of atoms. In general, strong or polar bonds are the result of the electron cloud of one atom being drawn towards the electron cloud of another atom, such that the negative or positive charge of the electron clouds increases. These behaviors merge into each other seamlessly in various circumstances, so that there is no clear line to be drawn between them. 
A chemical bond is an attraction between atoms. Many types of bond exist between atoms, such as single, double, and triple bonds and bonds of particular strength, including ionic and covalent bonds. The strength of chemical bonds between atoms ranges from the weakest to the strongest. The bond may result from the electrostatic force of attraction between opposite charges, dipole–dipole attraction (see also polarization (chemistry)), or the sharing of electrons as in covalent bonding. In chemistry, a chemical bond is a lasting attraction between atoms that enables the formation of chemical compounds. In general, strong or polar bonds are the result of the electron cloud of one atom being drawn towards the electron cloud of another atom, such that the negative or positive charge of the electron clouds increases  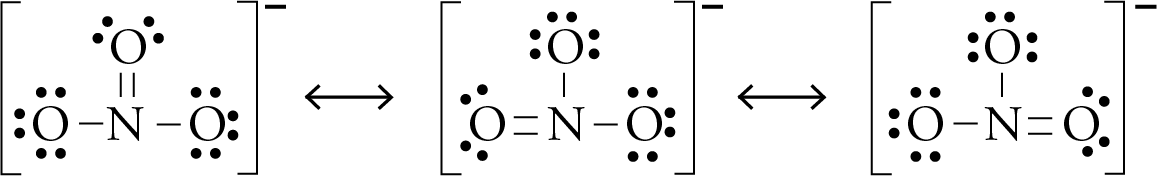 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
